|
12/23/2023 0 Comments Water viscosity versus temperature 
1 through 3 may be used to approximate water viscosity.įig. The 28 tables generated from the correlating equations cover :įigs. Their correlating equations involve 32 parameters for calculating the numerical effect of pressure, temperature, and concentration of aqueous NaCl solutions on the dynamic and kinematic viscosity of water. In the physical chemistry literature, there is an enormous amount of indirect evidence to support this concept.įor the best estimation of the viscosity of water, refer to Kestin et al. However, this effect is very small and has not been measured to date. In water, the presence of the gas actually causes the water molecules to interact with each other more strongly, thus increasing the rigidity and viscosity of the water. Gas-in-the-water phase behaves entirely differently than gas in hydrocarbons (personal communication with J.C. There is little information on the actual numerical effect of dissolved gas on water viscosity. In general, brine viscosity increases with: ĭissolved gas in the formation water at reservoir conditions generally results in a negligible effect on water viscosity. The viscosity of formation water, μ w, is a function of: This new technology promises to improve the ability to troubleshoot problems by directly measuring IFT instead of trial-and-error testing. IFT is a critical property of produced water, but is rarely measured because of the analytical difficulties. Computerized commercial pendant-drop and falling-drop tensiometers are now available for use by chemists in more general field R&D laboratories. The laboratory measurements traditionally have been difficult and done only by specialized facilities. Surface tension is measured in the laboratory by a tensiometer, by the drop method, or by a variety of other methods. 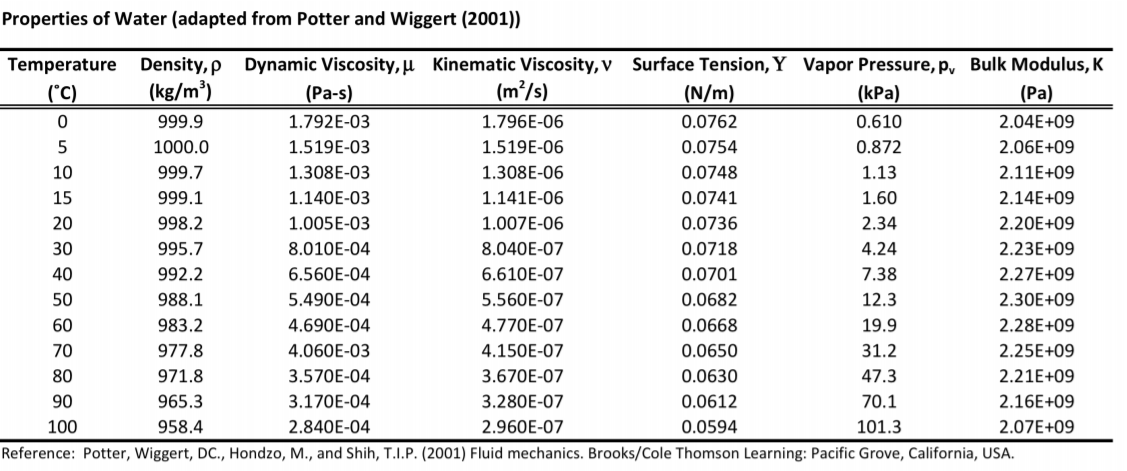
Formulating, selecting, testing, and troubleshooting emulsion breakers is the focus of an enormous amount of the effort devoted to the impacts of producing water with hydrocarbons. In attempting to separate the oil from the three-phase production stream, the addition of emulsion breakers changes the IFT and promotes the agglomeration of small droplets into larger ones that separate quickly. Indeed, certain corrosion inhibitors added to the three-phase production stream can lower the produced water IFT enough (<1 to 5 dyne/cm) to cause the droplet size of the entrained oil to be small enough that no injection well plugging is observed, even at high oil carryover (percent levels) in the reinjected produced water. Most chemicals added during the course of drilling or production have a major effect on the IFT of the produced water and the hydrocarbons. Also, the IFT determines the ease of separation of oil from water, because it determines the size of the oil or water droplets, depending on which phase is internal. IFT is an important factor in enhanced recovery. At very low values of IFT, oil and water become miscible and behave as a single phase. The lower the IFT, the smaller the droplet of the internal phase. If the phase boundary separates a liquid and a gas or a liquid and a solid, the attractive force at the boundary usually is called surface tension however, the attractive force at the interface between two liquids is called interfacial tension (IFT). Surface tension (interfacial tension or IFT) is a measure of the attractive force acting at a boundary between two phases. When resistivity of formation water is used in electric-log interpretation, the value is adjusted to formation temperature. Formation water resistivity, R wg, is expressed in units of Ω-m. The direct measurement method is essentially the electrical resistance through a l-m 2 cross-sectional area of 1 m 3 of formation water. It can be measured directly or calculated. O) hydrogen-bond.The resistivity of formation water is a measure of the resistance offered by the water to electrical current. Consult the literature (an internet source will do) and find an approximate strength for a typical (O-H.What is the value of \(E_a\) relative to ambient thermal kinetic energy (given by \(Ek = RT\))? Report the activation energy for viscous flow (\(E_a\)) for each liquid in units of kJ/mol and also rank them from smallest to largest.Accurately represent the temperature variation of viscosity for these liquids (note any discrepancies between your data and the best-fit line)? Does the model work better for any one of these liquids than it does for another? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
